The Waters Of The Ocean
-All matter is made of atoms
-Substances composed of 2 or more of the same atom are elements
-2 or more atoms combined is a molecule
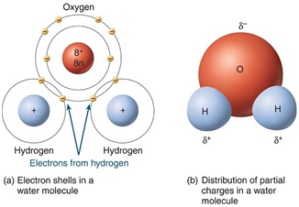
-Hydrogen bonds are the bonds that connect water molecules, hydrogen bonds to oxygen
-Water exists in 3 states: Liquid, solid, and gas
-As water cools, the molecules slow and the water becomes denser
-The amount of heat required to melt a substance is its latent heat of melting
-The amount of heat needed to raise a substance's temperature by a given amount is its heat capacity
-Water absorbs a great deal of heat when it evaporates
-Water is especially good at dissolving salts
-Ions- electrically charged atoms
-Solids dissolved in seawater enter the water through: rain, runoff/rivers/streams, and hydrothermal vents
-Seawater contains mostly solutes, or dissolved materials
-Salinity-The total amount of salt dissolved in seawater
-The percentage of major ions in seawater remains relatively constant all the time, know as the rule of constant proportions
-Precipitation- rain and snow
-Average salinity of the ocean is 35%
-A graph that shows the temperature at different depths in the ocean is called a temperature profile
-Water column- a temperature profile obtained from a given location cab be thought of as showing the temperature in a vertical shaft
-CTDs- Conductivity Temperature Depth meters
-Gases are also dissolved in seawater, the most important being: oxygen, carbon dioxide, and nitrogen
-Gas exchange occurs when gas goes from the ocean to the atmosphere and vise versa
-Photosynthesis and cellular respiration affect the amount of oxygen in the water
-Light easily penetrates water due to its transparency
-Substances composed of 2 or more of the same atom are elements
-2 or more atoms combined is a molecule
-Hydrogen bonds are the bonds that connect water molecules, hydrogen bonds to oxygen
-Water exists in 3 states: Liquid, solid, and gas
-As water cools, the molecules slow and the water becomes denser
-The amount of heat required to melt a substance is its latent heat of melting
-The amount of heat needed to raise a substance's temperature by a given amount is its heat capacity
-Water absorbs a great deal of heat when it evaporates
-Water is especially good at dissolving salts
-Ions- electrically charged atoms
-Solids dissolved in seawater enter the water through: rain, runoff/rivers/streams, and hydrothermal vents
-Seawater contains mostly solutes, or dissolved materials
-Salinity-The total amount of salt dissolved in seawater
-The percentage of major ions in seawater remains relatively constant all the time, know as the rule of constant proportions
-Precipitation- rain and snow
-Average salinity of the ocean is 35%
-A graph that shows the temperature at different depths in the ocean is called a temperature profile
-Water column- a temperature profile obtained from a given location cab be thought of as showing the temperature in a vertical shaft
-CTDs- Conductivity Temperature Depth meters
-Gases are also dissolved in seawater, the most important being: oxygen, carbon dioxide, and nitrogen
-Gas exchange occurs when gas goes from the ocean to the atmosphere and vise versa
-Photosynthesis and cellular respiration affect the amount of oxygen in the water
-Light easily penetrates water due to its transparency
- Different colors of light penetrate different depths of the ocean
- Violet goes nearly 98 m down
- Blue extends 197 m down
- Green goes 131 m down
- Yellow is 98 m down
- Orange is 42 m down
- Red is 33 m down
- Most important gases in the ocean are Oxygen, Carbon dioxide, and nitrogen
- The Amount of oxygen in the water also strongly affects the photosynthesis and respiration of organisms
- Each 10 meters you descend into the water the pressure will gain 1 ATM
- The Coriolis Effect is the rotation of the earth and the wind patterns and ocean currents effected by the effect!
- The trade winds are from the affect of warm air rising and cooling off so that the cool air pushed down towards the surface at a line across the equator
- we have 5 major winds that cycle in the worlds oceans, the largest being north of the Pacific
- The ocean consists of 3 layers firs being the upper layer consisting of 100 to 200 m down usually affected by wind
- thermocline's are when the temperature changes dramatically in the depth of water
- The intermediate layer is 1000 to 5000 m down
- and the Deep layer is usually 1500 m to the bottom of the sea floor
Waves and Tides
*Waves- wind not only drives surface
-crest the highest part of the wave
-trough the lowest part of a wave
-height the size of an ocean wave is usually expressed
-wavelength period the distance between crest or troughs
-seas stretched out waves
-swells form once waves get away from wind
-surf a wave pile up that steeps over creates this
*Tides- surface has been rising and falling in this rhythmic pattern
-tidal range difference in water level between areas
-mixed semidiurnal tide have successive high tides of different height
-neap tides when the sun and moon are at right angles their effects of partially cancel each other
-semidiural tides two high tides and two low tides a day
-Diurnal tides occur when there is only one high and one low tides every day
-tide tables that predict the time and height of high and low tides